BD-14775


- Overview
- Products & Accessories
- EIFU & Resources
- FAQ
A consistent, reproducible technique
The Echo 2™ Positioning System is a deployment and positioning device that comes attached to Ventralight™ ST Mesh. It facilitates mesh positioning and centering over the hernia defect, for a consistent, reproducible technique.
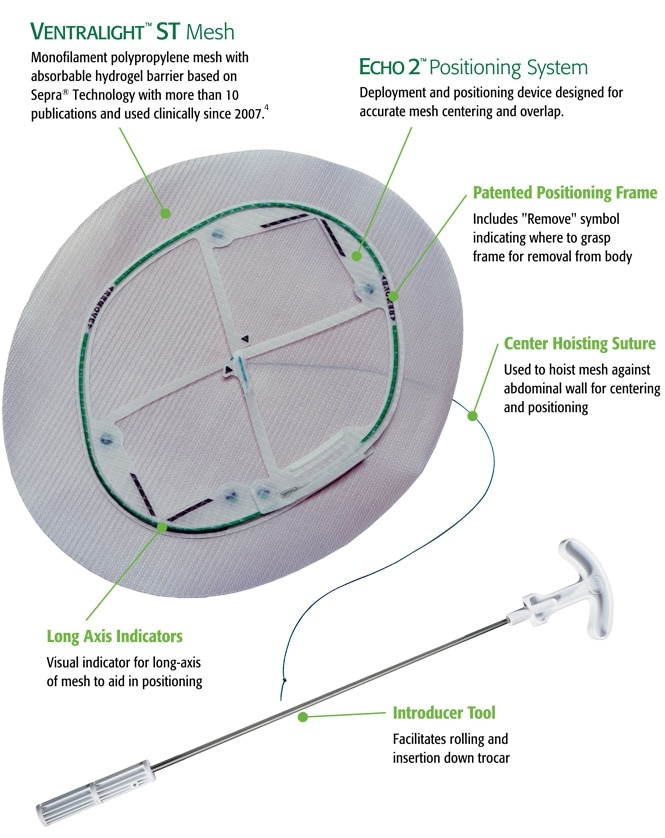
The Echo 2™ Positioning System is intended to aid with:
- Accurate mesh placement, positioning, and overlap1,3
- Decreasing operative time 1,3
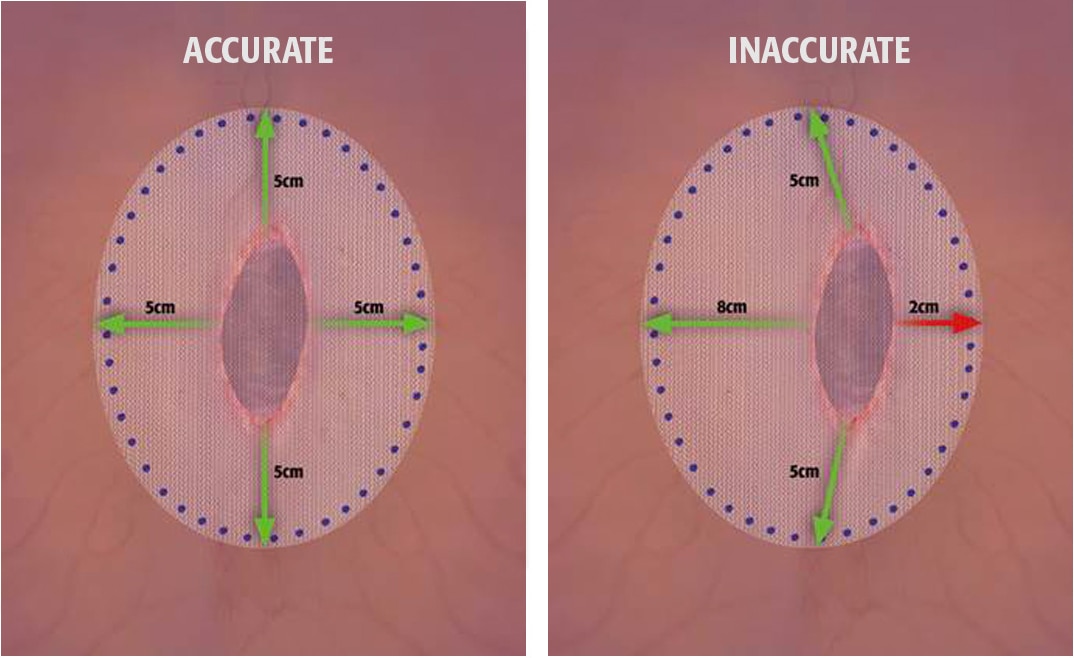
Literature suggests:
- Accurately centered mesh with appropriate overlap has shown to reduce the risk of mesh shift and recurrence 1,2
- Inaccurate mesh overlap in laparoscopic ventral hernia repair can result in postoperative mesh shift and recurrence 1,2
1 LeBlanc K. “Proper mesh overlap is a key determinant in hernia recurrence following laparoscopic ventral and incisional hernia repair“ Hernia 2016 Feb;20(1):85-99.
2 Liang MK, Clapp ML, Garcia A, Subramanian A, Awad SS. “Mesh shift following laparoscopic ventral hernia repair.” J Surg Res. 2012 Sep;177(1):e7-13.
3 Tollens T, Topal H, Ovaere S, Beunis A, Vermeiren K, Aelvoet C. “Prospective analysis of ventral hernia repair using the Ventralight™ ST hernia patch.” Surg Technol Int. 2013 Sep;23:113-6.
4 References available upon request.
Indications
Ventralight™ ST Mesh is indicated for use in the reconstruction of soft tissue deficiencies, such as for the repair of hernias. The Echo 2™ Positioning System is intended to facilitate the delivery and positioning of the soft tissue prosthesis during laparoscopic hernia repair.
Contraindications
Do not use the device in infants, children or pregnant women, whereby future growth will be compromised by use of such material. Do not use for the reconstruction of cardiovascular defects. Literature reports there is a possibility for adhesion formation when the polypropylene is placed in direct contact with the bowel or viscera.
Warnings
The use of any synthetic mesh in a contaminated or infected wound can lead to fistula formation and/or extrusion of the mesh and is not recommended. If an infection develops, treat the infection aggressively. Consideration should be given regarding the need to remove the mesh. Unresolved infection may require removal of the mesh Ventralight™ ST Mesh is the only permanent implant component of the device. The Echo 2™ Positioning System (which includes deployment frame, center hoisting suture and all connectors) must be removed from the patient and appropriately discarded. It is not part of the permanent implant. Do not apply sharp, pointed, cautery devices, or ultrasonic tools (such as scissors, needles, tackers, diathermic tools, etc.) to the Echo 2™ Positioning System frame. The device contains superelastic nitinol wire; do not cut and avoid direct contact/coupling with active surgical electrodes. The Echo 2™ Positioning System should not be used with any other hernia prosthesis aside from those with which it comes pre-attached/packaged.
Precautions
Do not trim the mesh. This will affect the interface between the mesh and the positioning system. Visualization must be maintained throughout the course of the entire surgical procedure. Additionally, laparoscopic removal of the Echo 2™ Positioning System frame must be performed under sufficient visualization of the entire device and surrounding anatomy, to ensure proper removal.
Adverse Reactions
Possible complications may include, but are not limited to, seroma, adhesion, hematoma, pain, infection, inflammation, extrusion, erosion, migration, fistula formation, allergic reaction and recurrence of the hernia or soft tissue defect.
Please consult package insert for more detailed safety information and instructions for use.
1 LeBlanc K. “Proper mesh overlap is a key determinant in hernia recurrence following laparoscopic ventral and incisional hernia repair“ Hernia 2016 Feb;20(1):85-99.
2 Liang MK, Clapp ML, Garcia A, Subramanian A, Awad SS. “Mesh shift following laparoscopic ventral hernia repair.” J Surg Res. 2012 Sep;177(1):e7-13.
3 Tollens T, Topal H, Ovaere S, Beunis A, Vermeiren K, Aelvoet C. “Prospective analysis of ventral hernia repair using the Ventralight™ ST hernia patch.” Surg Technol Int. 2013 Sep;23:113-6.
4 References available upon request.
Indications
Ventralight™ ST Mesh is indicated for use in the reconstruction of soft tissue deficiencies, such as for the repair of hernias. The Echo 2™ Positioning System is intended to facilitate the delivery and positioning of the soft tissue prosthesis during laparoscopic hernia repair.
Contraindications
Do not use the device in infants, children or pregnant women, whereby future growth will be compromised by use of such material. Do not use for the reconstruction of cardiovascular defects. Literature reports there is a possibility for adhesion formation when the polypropylene is placed in direct contact with the bowel or viscera.
Warnings
The use of any synthetic mesh in a contaminated or infected wound can lead to fistula formation and/or extrusion of the mesh and is not recommended. If an infection develops, treat the infection aggressively. Consideration should be given regarding the need to remove the mesh. Unresolved infection may require removal of the mesh Ventralight™ ST Mesh is the only permanent implant component of the device. The Echo 2™ Positioning System (which includes deployment frame, center hoisting suture and all connectors) must be removed from the patient and appropriately discarded. It is not part of the permanent implant. Do not apply sharp, pointed, cautery devices, or ultrasonic tools (such as scissors, needles, tackers, diathermic tools, etc.) to the Echo 2™ Positioning System frame. The device contains superelastic nitinol wire; do not cut and avoid direct contact/coupling with active surgical electrodes. The Echo 2™ Positioning System should not be used with any other hernia prosthesis aside from those with which it comes pre-attached/packaged.
Precautions
Do not trim the mesh. This will affect the interface between the mesh and the positioning system. Visualization must be maintained throughout the course of the entire surgical procedure. Additionally, laparoscopic removal of the Echo 2™ Positioning System frame must be performed under sufficient visualization of the entire device and surrounding anatomy, to ensure proper removal.
Adverse Reactions
Possible complications may include, but are not limited to, seroma, adhesion, hematoma, pain, infection, inflammation, extrusion, erosion, migration, fistula formation, allergic reaction and recurrence of the hernia or soft tissue defect.
Please consult package insert for more detailed safety information and instructions for use.
BD offers training resources to help improve your clinical practices as part of our goal of advancing the world of health.
BD supports the healthcare industry with market-leading products and services that aim to improve care while lowering costs. We host and take part in events that excel in advancing the world of health™.
BD promotes clinical excellence by providing various resources on best practices, clinical innovations and industry trends in healthcare.
1 LeBlanc K. “Proper mesh overlap is a key determinant in hernia recurrence following laparoscopic ventral and incisional hernia repair“ Hernia 2016 Feb;20(1):85-99.
2 Liang MK, Clapp ML, Garcia A, Subramanian A, Awad SS. “Mesh shift following laparoscopic ventral hernia repair.” J Surg Res. 2012 Sep;177(1):e7-13.
3 Tollens T, Topal H, Ovaere S, Beunis A, Vermeiren K, Aelvoet C. “Prospective analysis of ventral hernia repair using the Ventralight™ ST hernia patch.” Surg Technol Int. 2013 Sep;23:113-6.
4 References available upon request.
Indications
Ventralight™ ST Mesh is indicated for use in the reconstruction of soft tissue deficiencies, such as for the repair of hernias. The Echo 2™ Positioning System is intended to facilitate the delivery and positioning of the soft tissue prosthesis during laparoscopic hernia repair.
Contraindications
Do not use the device in infants, children or pregnant women, whereby future growth will be compromised by use of such material. Do not use for the reconstruction of cardiovascular defects. Literature reports there is a possibility for adhesion formation when the polypropylene is placed in direct contact with the bowel or viscera.
Warnings
The use of any synthetic mesh in a contaminated or infected wound can lead to fistula formation and/or extrusion of the mesh and is not recommended. If an infection develops, treat the infection aggressively. Consideration should be given regarding the need to remove the mesh. Unresolved infection may require removal of the mesh Ventralight™ ST Mesh is the only permanent implant component of the device. The Echo 2™ Positioning System (which includes deployment frame, center hoisting suture and all connectors) must be removed from the patient and appropriately discarded. It is not part of the permanent implant. Do not apply sharp, pointed, cautery devices, or ultrasonic tools (such as scissors, needles, tackers, diathermic tools, etc.) to the Echo 2™ Positioning System frame. The device contains superelastic nitinol wire; do not cut and avoid direct contact/coupling with active surgical electrodes. The Echo 2™ Positioning System should not be used with any other hernia prosthesis aside from those with which it comes pre-attached/packaged.
Precautions
Do not trim the mesh. This will affect the interface between the mesh and the positioning system. Visualization must be maintained throughout the course of the entire surgical procedure. Additionally, laparoscopic removal of the Echo 2™ Positioning System frame must be performed under sufficient visualization of the entire device and surrounding anatomy, to ensure proper removal.
Adverse Reactions
Possible complications may include, but are not limited to, seroma, adhesion, hematoma, pain, infection, inflammation, extrusion, erosion, migration, fistula formation, allergic reaction and recurrence of the hernia or soft tissue defect.
Please consult package insert for more detailed safety information and instructions for use.
